Electrochemistry and Plastics: A Key Combination for a Sustainable Future

Electrochemistry studies the transformation of electricity into chemical reactions and, conversely, how certain chemical reactions can generate electricity. What’s most interesting is that electricity can be regulated with great precision (potential, intensity, time), making it a key tool for developing more efficient, sustainable, and selective processes, enabling innovative solutions that were previously unthinkable. Thanks to this, electrochemistry is used not only to generate or store energy but also to create new materials, recover resources, or drive cleaner processes across various industrial sectors.
In this way, electrochemistry is expanding its reach to many other materials and sectors. One of the most promising is plastics, where a wide range of opportunities is opening up for innovation in sustainability, recycling, and new functional products.
How is electrochemistry related to plastics?
At first glance, electrochemistry and plastics seem to belong to very different worlds: one is associated with metals and electricity, and the other with lightweight, insulating materials. However, they are increasingly connected in the development of advanced and sustainable technologies. Electrochemistry allows chemical reactions to be controlled through electricity, while plastics offer flexibility, lightness, and stability. Together, they form the foundation of many emerging devices and processes.
In the plastics world, electrochemistry enables material recovery, production of high-value compounds that can act as polymerization initiators, surface modification, generation of functional films, or even the creation of new hybrid materials through techniques such as electropolymerization.
Moreover, the relationship between plastics and electrochemistry is bidirectional: not only does electrochemistry play an important role in the plastics industry, but plastics also enable new electrochemical functions. Thanks to their flexibility, permeability, and chemical stability, polymers make it possible to develop lightweight, safe, and versatile devices that could not previously be designed using purely metallic materials.
A clear example is lithium-ion batteries. Although we often see them as “metallic” devices, polymers play essential roles inside them: polymer separators prevent short circuits and maintain selective ion flow; conductive membranes or polymer gels enable lithium ion transport while maintaining device safety; and plastic binders and coatings contribute to electrode stability and durability.
In other words, without these functional plastics, today’s batteries would not be safe, stable, or efficient. In turn, electrochemistry drives the creation of new conductive polymers and advanced membranes, thus closing the innovation loop.

Parts of a lithium-ion battery.
Real Applications of Electrochemistry in the Plastics World
Electrochemistry enables innovative use of plastics in several areas. Some of the most noteworthy applications include:
1. Electrochemical Recycling and Depolymerization
- Enables breaking polymer bonds and recovering monomers or valuable chemicals.
- Examples: decomposition of polyolefins (such as polyethylene and polypropylene) and polyesters (such as polyethylene terephthalate).
- This electrochemical chemical recycling is more efficient, generates less waste, and reduces the need for heat or harsh chemicals.
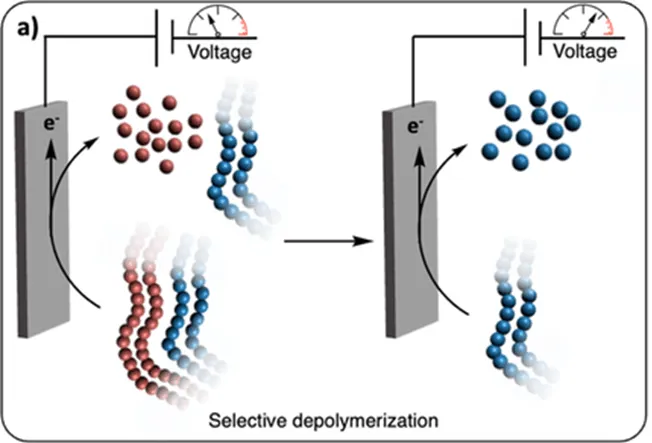
Electrochemical depolymerization. Image adapted from the article by Zhang et al. Chemical Science 15, 2024 (DOI: 10.1039/D4SC01754D).
2. CO₂ Conversion into Plastic Precursors
- Electrochemistry can reduce CO₂ into molecules such as CO, ethylene, formic acid, or methanol.
- These compounds are used as building blocks for sustainable plastics, closing the material cycle and reducing dependence on fossil resources.
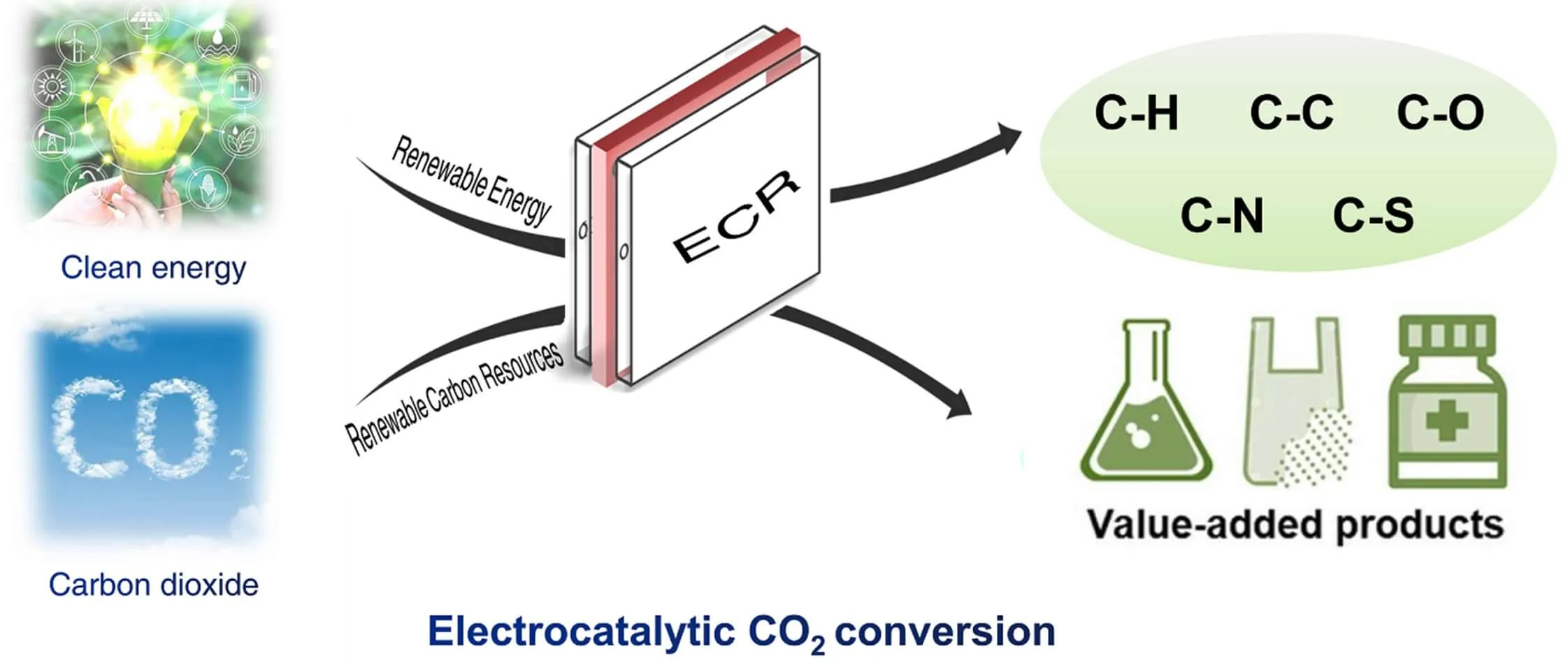
Electroconversion of CO₂ into high-value chemical compounds.
3. Functional Plastics in Electrochemical Technologies
- In batteries, supercapacitors, or fuel cells, plastics play an active role as:
- Separators and membranes that allow ion flow but block electrons.
- Coatings or solid electrolytes that provide stability, safety, and durability.
- These materials show that plastics are not only transformed or recycled but also facilitate the evolution of electrochemistry, enabling the development of more efficient and innovative technologies.
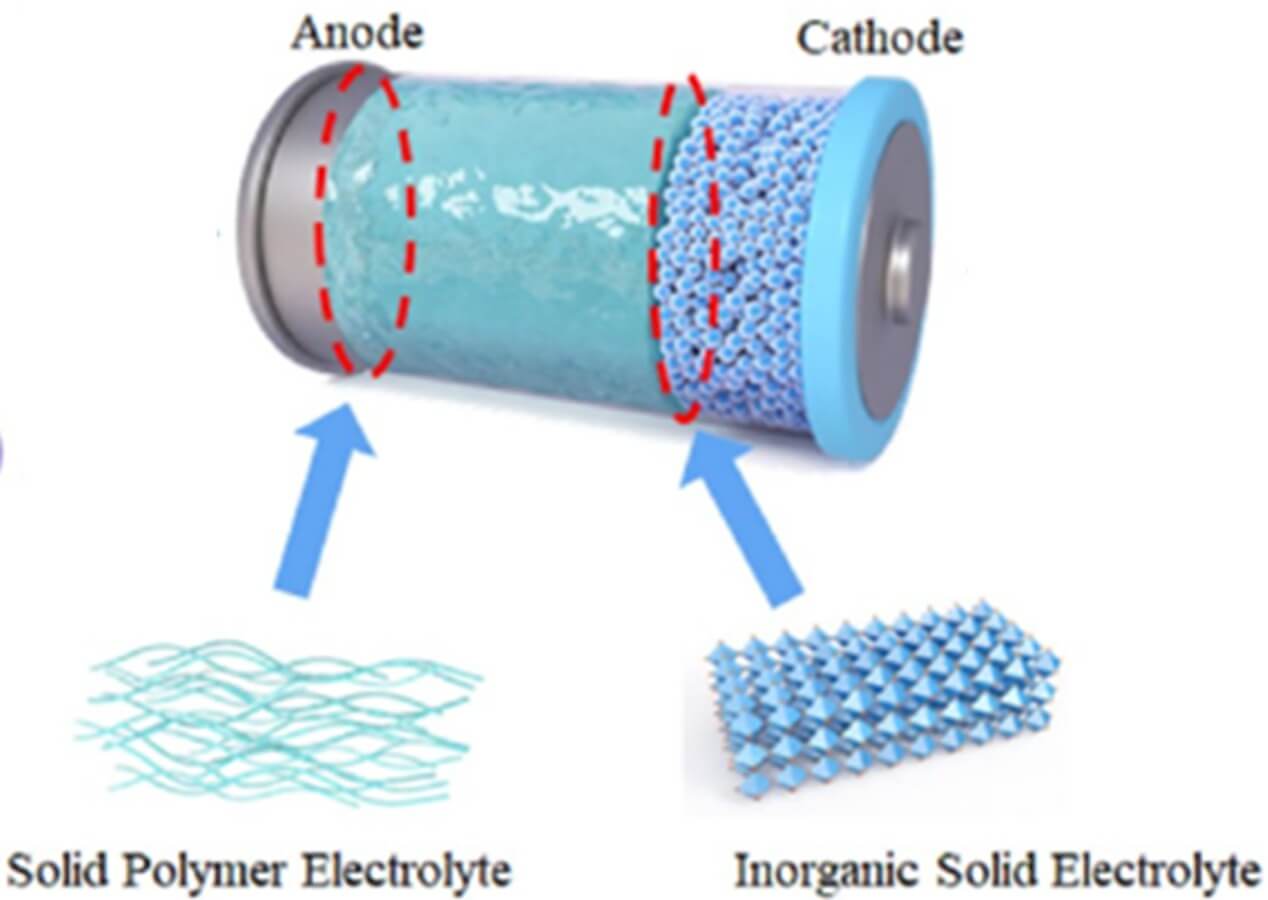
Lithium-ion battery with a polymer ion-gel electrolyte. Photo adapted from Yang et al. Nano-Micro Letters 15, 74, 2023 (DOI: 10.1007/s40820-023-01051-3).
4. Sensors and Smart Packaging
- Conductive polymers or special coatings are integrated into electrochemical sensors capable of detecting gases, pH, or chemical changes.
- Applications: food packaging that monitors freshness, medical devices, or environmental sensors.
- Plastics provide flexibility and chemical compatibility, while electrochemistry enables precise and reliable detection.
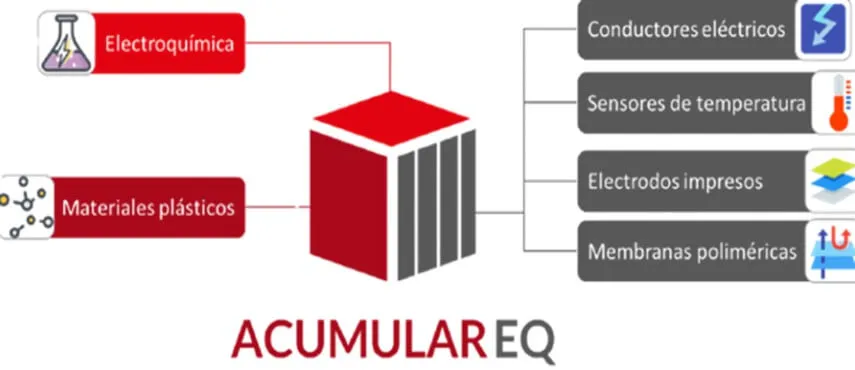
Schematic summary of the IVACE project “ACUMULAR EQ” developed at AIMPLAS, where plastic materials are combined with electrochemistry to develop temperature sensors, printed electrodes, and polymer membranes.
Advantages of Electrochemistry in the Plastics Industry
From all the applications above, it’s clear that electrochemistry not only offers new ways to process, recycle, and functionalize plastics but also provides cross-cutting advantages that are transforming how technology is developed.
As a process controlled by electricity, it allows for high precision, reduced chemical consumption, operation at lower temperatures and pressures, and the development of more durable, efficient, and sustainable materials.
These advantages, summarized in the following image, explain why electrochemistry is becoming a key tool in the development of polymeric materials and in the path toward a greener industry.

What Can AIMPLAS Offer?
AIMPLAS has the experience and infrastructure needed to drive electrochemistry into the plastics sector:
- Material and catalyst development: manufacturing electrodes, electrolytes, and catalysts tailored to each type of polymer or application.
- Process optimization: testing to scale electrochemical technologies from lab to pilot plant.
- Advanced characterization: surface analysis, chemical properties, and electrochemical performance of materials.
- Participation in innovation projects: national and European, working alongside companies and technology centers.
Electrochemistry at AIMPLAS: a key tool for a more efficient and circular plastics sector
Electrochemistry and plastics are more connected than they seem. Electrochemistry not only enables the recycling and transformation of polymers or the production of high-value compounds that can initiate polymerization, but plastics also facilitate the development of advanced electrochemical technologies such as batteries, sensors, or smart packaging.
AIMPLAS combines electrochemistry with different fields, such as catalysis and membranes, applying a cross-disciplinary approach that drives innovative and sustainable solutions. In this way, electrochemistry becomes a key tool for advancing toward a more efficient, functional, and environmentally friendly plastics sector.