Biological evaluation and biocompatibility of medical devices
The evaluation of oxidative stress through bioassays is essential to understand its effects and ensure the safety of the consumer and the environment.
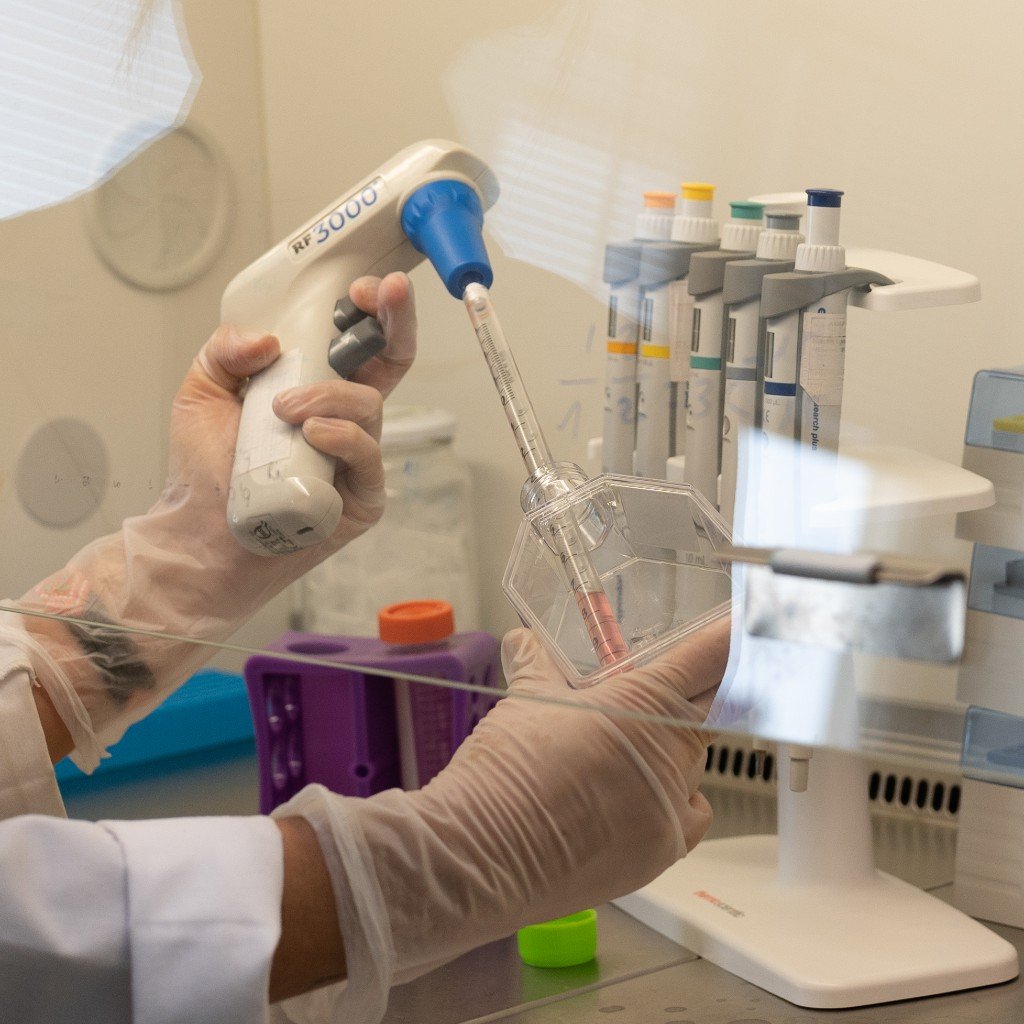
Ensure the safety and compliance of your medical devices with AIMPLAS’ new cell culture laboratory.
The medical device market is evolving rapidly. Innovation is essential, but so is ensuring patient safety and regulatory compliance.
At AIMPLAS, we have a new cell culture laboratory specialised in the biological evaluation and biocompatibility of medical devices in accordance with Regulation (EU) 2017/745 and UNE-EN ISO 10993 standards.
What do we offer?
A comprehensive preclinical and biocompatibility assessment service that enables you to:
- Determine the biological response of materials and medical devices.
- Validate the safety of products such as face masks, gloves, catheters, electrodes, syringes, dressings or implants.
- Accelerate CE marking, an essential requirement for marketing in the EU.
- Replace in vivo tests with in vitro bioassays, in line with the 3Rs principles (replace, reduce and refine the use of animals).
Available tests
Our laboratory performs studies under ISO/IEC 17025 and Good Laboratory Practice (GLP) conditions, including:
- Cytotoxicity (UNE-EN ISO 10993-5).
- Skin irritation and sensitisation (UNE-EN ISO 10993-10).
- Systemic toxicity and genotoxicity (UNE-EN ISO 10993-11 and 10993-3).
- Blood and tissue interaction (UNE-EN ISO 10993-4).
Why AIMPLAS?
- Multidisciplinary expertise: Our experience across the value chain allows us to support you throughout the material synthesis, manufacturing and validation process.
- Personalised technical advice: We focus on guidance, design and consultancy to offer the most suitable strategy for each material.
- Comprehensive regulatory approach: Thanks to our multidisciplinary team, we work closely with other AIMPLAS departments to meet all regulatory requirements and obtain the necessary CE marking for medical materials.
- Commitment to responsible innovation: We support the development of safer and more sustainable medical products, actively collaborating in innovation projects with companies in the sector.
Ensure the compliance and safety of your medical devices. Contact AIMPLAS and let our team guide you through the biological validation of your medical products.