Cleanrooms in the pharmaceutical industry: what they are and why they matter

Cleanrooms are a critical element in the healthcare sector when working on high value-added applications such as pharmaceutical compounding. Technologies such as Hot Melt Extrusion (HME) and Twin-Screw Melt Granulation (TSMG) require controlled environments where contamination is strictly limited.
In these processes, material quality depends not only on the formulation or the technology, but also on the environment. For this reason, cleanrooms have become a key requirement to ensure safety, regulatory compliance and reproducibility.
What is a cleanroom and why is it key in pharmaceutical compounding?
A cleanroom is an environment in which critical variables are precisely controlled, such as:
- Airborne particles: managed through air filtration systems and particle counting. Values determine air cleanliness according to the limits set in ISO 14644-1.
- Microbiological contamination: controlling environmental conditions and particles significantly reduces the presence of microorganisms. In addition, cleanrooms typically implement specific cleaning and sterilisation procedures.
- Temperature and humidity: controlled using heating and cooling systems. Values depend on process and/or material requirements, although typical conditions are around 21 ± 2°C and 60 ± 5% RH, respectively.
- Differential pressure between rooms: controlled through regulated air supply/return systems and airlock doors. Values depend largely on facility size and layout. This prevents unwanted particle migration between areas.
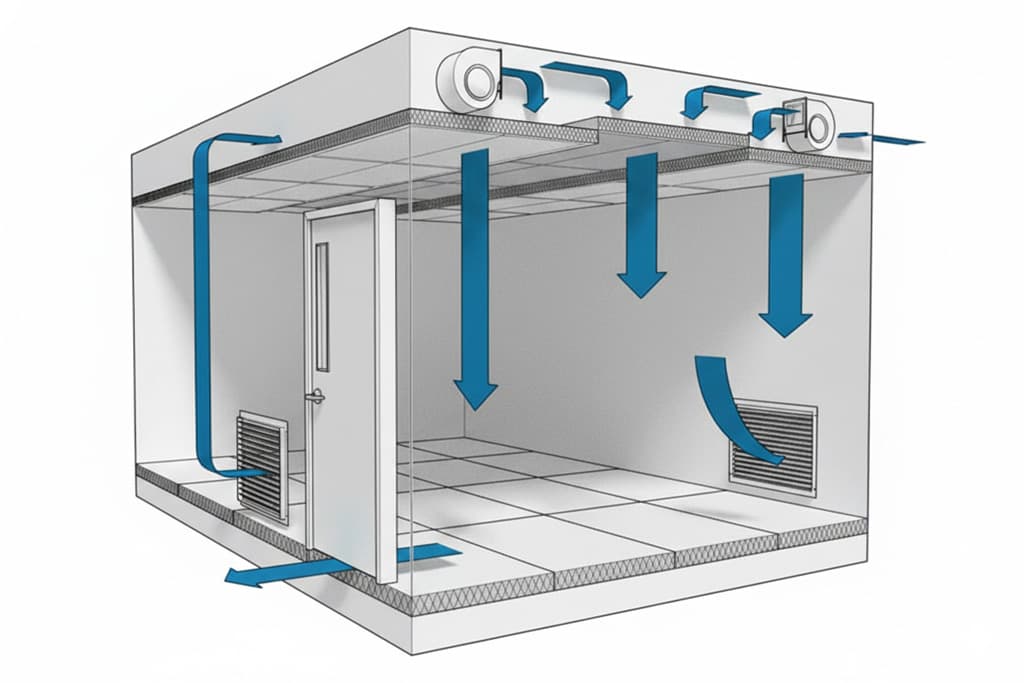
In the context of HME and TSMG, these factors directly impact:
- API (Active Pharmaceutical Ingredient) stability: APIs are often highly sensitive to humidity and temperature, with specific storage and processing ranges.
- Blend homogeneity: can be affected if raw materials have undergone changes or if there are variations in key process stages.
- Final product safety: a controlled environment ensures the absence of foreign matter or contaminants during manufacturing.
Strict control of environmental and working conditions prevents changes in compounds, ensures reproducibility, avoids cross-contamination and guarantees compliance with regulatory requirements.
Cleanroom classification according to ISO 14644
The ISO 14644 standard defines air cleanliness levels. In pharmaceutical plastics processing, selecting the appropriate class is essential to balance quality and cost.
| ISO Class | Max. particles ≥0.5 µm (particles/m³) | Stringency level | Typical application in HME / TSMG |
|---|---|---|---|
| ISO 1 | 10 | Extremely high | Advanced R&D for critical formulations |
| ISO 2 | 100 | Ultra high | High-precision pharmaceutical microstructures |
| ISO 3 | 1,000 | Very high | Highly sensitive development processes |
| ISO 4 | 10,000 | Very high | Scale-up of critical formulations |
| ISO 5 | 3,520 | Very high | Sterile production / highly sensitive APIs |
| ISO 6 | 35,200 | High | Controlled pharmaceutical processes |
| ISO 7 | 352,000 | Medium-high | Standard HME / TSMG in GMP environments |
| ISO 8 | 3,520,000 | Medium | Plastic compounding with controlled requirements |
| ISO 9 | 35,200,000 | Low | Auxiliary or pre-processing areas |
The most common classes for HME and TSMG processes are ISO 7 and ISO 8, although ISO 5 and ISO 6 are required where critical stages or highly sensitive APIs are involved. The choice depends on product risk and the level of API exposure.
Good practices in cleanrooms for compounding
Strict control of working conditions (humidity, temperature and pressure) and ISO classification are not the only requirements for efficient and safe pharmaceutical compounding. Proper use of the cleanroom is just as important as its design.
Key good practices include:
- Strict access protocols
- Specific training in GMP environments
- Cleaning procedures adapted to polymer residues
- Continuous particle monitoring
- Periodic process validation
- Use of certified equipment for pharmaceutical manufacturing
These practices are framed within compliance with standards and regulations such as:
- ISO 14644: classification, design, validation and monitoring of controlled environments and cleanrooms
- ISO 14698: microbiological control in cleanrooms, including principles, methodology and monitoring of risk areas
- ISO 13485: design, production, installation and maintenance of medical devices, including polymer-based devices
- Good Manufacturing Practice (GMP): the mandatory basis for pharmaceutical production, defining cleanroom grades, particle limits, microbiological contamination and operating conditions, regulated by the EMA in Europe and the FDA in the United States
Cleanroom configurations
Cleanroom configuration is a key aspect in the design of controlled environments, especially in healthcare applications such as pharmaceutical compounding.
The main objective is to control incoming and outgoing air to limit particles, reduce contamination risks, protect the product and, where necessary, contain hazardous substances.
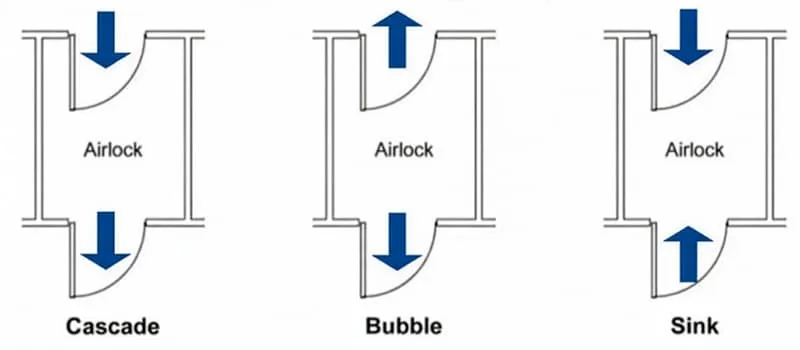
Cleanrooms typically consist of multiple rooms or zones. Airflow and pressure configuration can be:
- Cascade pressure: the most common in pharmaceutical environments. A positive pressure gradient is established between rooms, with air flowing from cleaner to less clean areas. This prevents contaminant ingress and protects the product.
- Bubble pressure: based on positive pressure in a specific area. A “bubble” of high pressure is created in a critical room, with air flowing outward in all directions. This offers maximum product protection and isolates the room from adjacent areas. Recommended for highly sensitive APIs or critical formulations.
- Sink (negative) pressure: the critical room operates at lower pressure than adjacent areas, causing air to flow into the room. This contains hazardous substances and protects both the environment and operators. Recommended for highly active or potentially hazardous APIs.
- Hybrid configuration: combines different approaches (e.g. cascade + bubble or cascade + sink). These are the most versatile, but also the most complex and costly to implement.
Projects carried out in the AIMPLAS Cleanroom
AIMPLAS has an ISO 7-certified cleanroom for the development of materials, compounds and devices produced via HME or TSMG under controlled, high-purity conditions. With a total area of 130 m², the facility includes HEPA air filtration systems and precise climate control for temperature and humidity. Rooms operate under positive pressure and are equipped with airlock doors to prevent external contamination. Separate personnel and material entry points minimise cross-contamination. Extrusion equipment is GMP-certified for pharmaceutical applications, ensuring strict traceability and thorough cleaning of all components.
AIMPLAS carries out numerous publicly and privately funded projects. Three notable examples include:
DEGLUMED

Focused on developing easy-to-swallow medicines and nutraceuticals using advanced pharmaceutical compounding technologies (HME).
Key aspects:
- Use of HME to disperse APIs in polymer matrices
- Development of innovative solid dosage forms (e.g. orodispersible or fast-release systems)
- Reduction or elimination of solvent use
ETERNAL
A European project (ID: 101057668) coordinated by AIMPLAS, aimed at sustainable pharmaceutical manufacturing, use and disposal.
Key aspects:
- Implementation of continuous twin-screw extrusion processes
- Optimisation of solvent-free granulation parameters
- Reduction of intermediate steps and improved energy efficiency
NUTRAFARM

Focused on polymer-based solutions for nutraceutical applications.
Key aspects:
- Design of polymer matrices for controlled release, suitable for 3D printing
- Compounding processes adapted to sensitive ingredients
- Taste masking, improved homogeneity and reproducibility
Do you need support in polymer-based pharmaceutical compounding?
Cleanrooms in the plastics sector are a key element for advanced processes such as HME and TSMG in pharmaceutical compounding. They not only ensure regulatory compliance, but also guarantee product quality, stability and reproducibility.
Proper environment design, implementation of best practices and regulatory compliance are essential to compete in an increasingly demanding market.
Having specialised expertise in polymer processing and pharmaceutical manufacturing in controlled environments helps reduce risks and accelerate the development of new products. Relying on experts in materials, processes and cleanroom technology is key to taking innovation to the next level.