Antimicrobial resistance and pharmaceutical manufacturing: the role of the SSbD framework
Antimicrobial resistance (AMR) is one of the foremost global health challenges of the 21st century. Among its contributing factors, effluents from the pharmaceutical industry are attracting growing attention. The Safe and Sustainable by Design (SSbD) framework, together with continuous manufacturing technologies such as Hot Melt Extrusion (HME) and Twin-Screw Granulation (TSG), makes it possible to reduce these residues by acting directly at the source of the production process.
What is antimicrobial resistance?
Antimicrobial resistance (AMR) is the ability of microorganisms to survive or even proliferate in the presence of drugs designed to inhibit or kill them. It is a critical global health threat, with 10 million deaths caused by AMR and 1 trillion US$ of additional healthcare costs projected by 2050. In addition, drug-resistant infections affect the health of animals and plants, reduce agricultural productivity, and threaten food security.
Antimicrobial resistance is primarily developed by microorganisms (such as bacteria, viruses, fungi, and parasites) through exposure to antimicrobial drugs (including antibiotics, antivirals, antifungals, and antiparasitics), which selects for resistant mutants or facilitates the acquisition of resistance genes.
What impact does the pharmaceutical industry have on AMR?
While overprescription and misuse of antibiotics are often in the spotlight as primary drivers, environmental exposure is increasingly recognised as a contributing factor.
The direct impact of pharmaceutical manufacturing on AMR prevalence has not yet been fully quantified; however, effluents from the pharmaceutical industry contain some of the highest concentrations of antibiotics and antibiotic resistance genes detected in the environment.
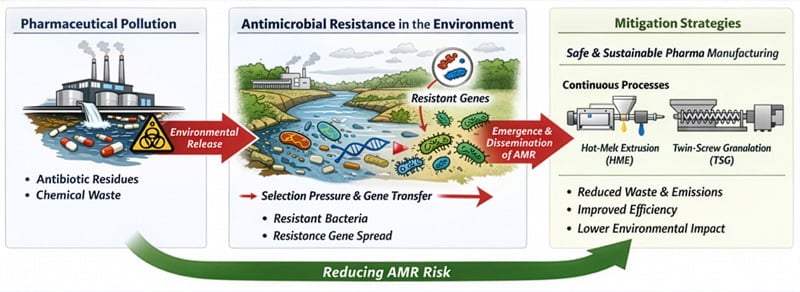
One of the main reasons is that traditional pharmaceutical manufacturing often relies on solvent-intensive operations —e.g., wet granulation, crystallisation—, generating high volumes of wastewater and solvent-containing effluents. When inadequately treated, these streams may contain residual active pharmaceutical ingredients at concentrations sufficient to exert biological activity.
Even sub-inhibitory concentrations in receiving environments (surface waters, sediments, and soils) can create selective pressure that favours the proliferation of resistance. The co-presence of other chemicals can exacerbate AMR by creating complex selective pressures that promote horizontal gene transfer and the survival of resistant microorganisms.
Additionally, there are currently no fully harmonised regulations that set specific limits for antimicrobials or broader pharmaceutical concentrations in industrial effluents such as those from pharmaceutical manufacturing. Regulatory frameworks traditionally focus on general pollutant levels (e.g., BOD, COD) or on controlling antimicrobial use, rather than directly regulating environmental discharges.
What is the SSbD framework and how does it reduce antibiotic contamination?
Implementing manufacturing processes that minimise waste is key to reducing environmental contamination and the selective pressure that derives from it. This approach is rooted in the Safe and Sustainable by Design (SSbD) framework, a set of principles which aims to integrate environmental safety and sustainability strategies across all stages of a drug lifecycle, from design to manufacturing to end-of-life disposal.
At the manufacturing stage, the emerging technologies that align with the SSbD paradigm and can replace waste-intensive batch processes are Hot Melt Extrusion (HME) and Twin-Screw Granulation (TSG).
HME and TSG: continuous manufacturing technologies without solvents
What is Hot Melt Extrusion (HME)?
Hot Melt Extrusion (HME) is a continuous manufacturing process in which drugs and excipients are melted and mixed using heat and mechanical shear to form a homogeneous solid dispersion, eliminating the need for solvents.
What is Twin-Screw Granulation (TSG)?
Twin-Screw Granulation (TSG) is a continuous granulation method where powders are mechanically mixed and agglomerated using twin screws, often with minimal or no liquid binder, producing uniform granules whilst reducing solvent use and downstream drying.
Advantages of HME and TSG over traditional processes:
- Reduced waste: these technologies can minimise or entirely replace solvent use through thermally and mechanically driven mixing.
- Enhanced process control: these manufacturing methodologies allow the integration of real-time monitoring using process analytical technology (PAT), which reduces batch failures and off-spec products and, in turn, decreases the volume of discarded materials.
- Lower energy intensity: continuous operations typically run at lower energy intensity per unit produced, reducing emissions and contributing to more sustainable and cost-efficient manufacturing.
Challenges and future outlook
Adopting these manufacturing processes is not without challenges. Technical hurdles include equipment investment, scale-up, and regulatory alignment. However, by adopting continuous manufacturing aligned with SSbD principles, pharmaceutical companies have a unique opportunity to tackle AMR at its source, making production more sustainable, efficient, and cost-effective.
How can AIMPLAS support the transition to SSbD pharmaceutical manufacturing?
AIMPLAS can support the shift away from traditional batch manufacturing towards more sustainable, continuous processing through applied research and pilot-scale infrastructure. By bridging the gap between research and industry, we facilitate the adoption of continuous manufacturing technologies through:
- Investigation at laboratory scale: using solid-state batch processes, we screen formulations that enable solvent-free or low-solvent manufacturing, reducing the potential generation of antibiotic-contaminated effluents.
- Evaluation of technical feasibility at pre-pilot scale: using microextruders (5–10 g/batch) and torque rheometers (30–300 g/batch), we optimise shear-driven processing conditions, supporting the transition away from solvent-based operations.
- Process validation under industrially relevant conditions: using twin-screw extruders in an ISO 7 cleanroom, we demonstrate scalable, continuous manufacturing routes.
Is your company evaluating the transition towards more sustainable manufacturing processes? Tell us where you are in the process and we will analyse together which AIMPLAS capabilities best fit your needs.
References
World Health Organization. (retrieved March 2026). Antimicrobial resistance. https://www.who.int/news-room/fact-sheets/detail/antimicrobial-resistance
World Health Organization. (2022). Global antimicrobial resistance and use surveillance system (GLASS) report: 2022. https://www.who.int/publications/i/item/9789240062702