Extrusion technology in the pharmaceutical sector
Extrusion is a manufacturing process developed in the early 1930s that has become a widely used technique in the food industry and, most notably, in the plastics industry. Years later, its potential was demonstrated in the pharmaceutical industry, especially in the formulation of solid dispersions and in the development of new and advanced dosage and drug delivery forms.
This technology addresses various challenges faced by the pharmaceutical industry, such as:
-
Poor drug bioavailability due to low solubility.
-
Low stability of the API (active pharmaceutical ingredient) during the manufacturing and processing of the medicine.
-
Unreliable sustained release.
-
Poor stability or tolerance of the API in the stomach.
-
Unpleasant taste of the medicine.
-
Limited film production.
These drug manufacturing processes have been designed to mix active ingredients with excipients (which must exhibit thermoplastic behaviour) for the development of such formulations. One of the main differences between conventional and pharmaceutical extrusion processes lies in the equipment used, as they must meet certain regulatory requirements: the parts of pharmaceutical extruders that come into contact with materials must not react with them or release components into the extruded product. These machines are designed to meet the cleaning and validation standards applicable to the pharmaceutical industry.
Pharmaceutical compounding processes
Pharmaceutical compounding processes include several specialised extrusion techniques:
-
Hot Melt Extrusion (HME): temperatures are applied up to the melting or softening point of thermoplastic materials to enable extrusion. This can be done using single-, twin-, or multi-screw extruders.
-
Twin Screw Extrusion (TSE): the material passes through two screws rotating either in the same or opposite directions to provide efficient mixing of the materials.
Extrusion applications
Amorphous Solid Dispersion (ASD)
This technique aims to enhance and optimise the solubility of certain active ingredients. These drugs are combined with a polymeric excipient that, when subjected to an extrusion process, results in the drug being dispersed in its amorphous form. As a result, solubility and absorption in the gastrointestinal system are improved. HME processes are used for these applications, where processing temperatures are applied above the polymer’s glass transition temperature but below the melting point of the API to ensure adequate stability and dispersion.
This technique enables pharmaceutical formulations with various functionalities:
-
Taste masking, since most APIs trigger an unpleasant taste.
-
Controlled release, in which the active ingredient is retained in a polymeric matrix, tailored to specific therapeutic needs.
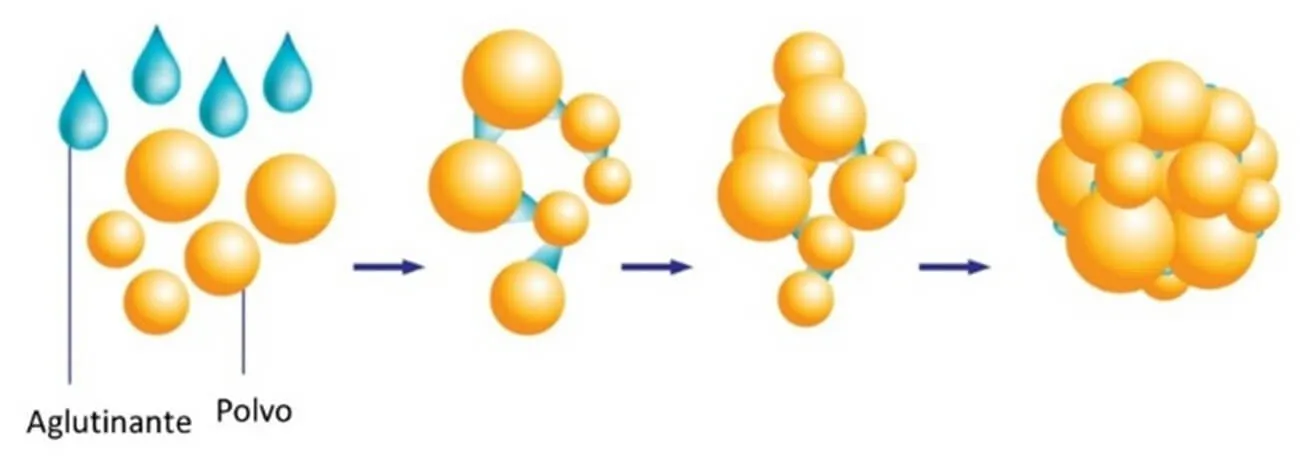
Granulation
One of the main issues when formulating pharmaceutical products, particularly in tablet form, is the low flowability of their ingredients, which makes it difficult to achieve proper compression. However, granules resulting from an extrusion process exhibit much greater flowability than the raw materials used, allowing for easy compression and resulting in a final product with improved compaction and suitable disintegration in the gastrointestinal tract. Additionally, the granules are composed of solid dispersions, which increase the solubility of active compounds and, therefore, their bioavailability.
Currently, AIMPLAS is carrying out the European project ETERNAL, aimed at optimising the granulation process of APIs on an industrial scale. To this end, granulation processes by melting or TSMG (Twin Screw Melt Granulation) are being implemented, resulting in granulated drug formulations by melting or softening binders. However, other granulation techniques also exist, such as wet granulation using liquid binders (usually water) or dry granulation, which requires neither liquids nor molten ingredients.
Films
These are drug delivery systems based on polymer films that incorporate pharmaceutical or nutraceutical active ingredients.
Different types of film formulations produced via extrusion include:
-
Mucoadhesive films: applied to adhere to the buccal or gingival mucosa, allowing either rapid or sustained release. Once treatment is completed or the active ingredient in the strip is exhausted, it must be removed.
-
Fast-dissolving films: strips that completely dissolve in the oral cavity within 60 seconds. They have a larger contact surface than conventional fast-dissolving tablets, resulting in better disintegration. These films are also highly flexible, which improves post-processing and transport. They can be presented as ODFs (orodispersible films) or sublingual strips.
DEGLUTECH is a Strategic Collaborative Project in the Valencian Community launched at AIMPLAS at the end of 2024. It involves developing formulations of various nutraceutical and pharmaceutical active ingredients in the form of fast-dissolving buccal films using HME processes, aimed at people with swallowing difficulties.
There is a wide variety of additional pharmaceutical compounding applications achieved through extrusion technologies, such as the formation of co-crystals, pharmaceutical salts, gels, microencapsulated compounds, solid lipid nanoparticles, implants, and abuse-deterrent formulations. Moreover, new areas remain to be explored, such as the use of extrusion in bioprinting processes.
Advantages of using extrusion in pharmaceutical applications
The use of extrusion techniques in the development of pharmaceutical applications offers numerous general advantages that address challenges posed by conventional manufacturing processes:
-
Continuous processing and ease of scaling up.
-
Integration of processes, resulting in faster, more efficient production with lower energy consumption compared to conventional methods.
-
Applicable to thermolabile or moisture-sensitive APIs due to the possibility of working under anhydrous and low-temperature conditions.
-
Improved API stability.
-
Sustainable techniques owing to reduced energy consumption and solvent use.
In conclusion, extrusion techniques offer an adaptable solution for the pharmaceutical industry, overcoming the limitations of conventional methods and improving the quality of pharmaceutical products.